
For questions or remarks please contact us. Limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all reagents. 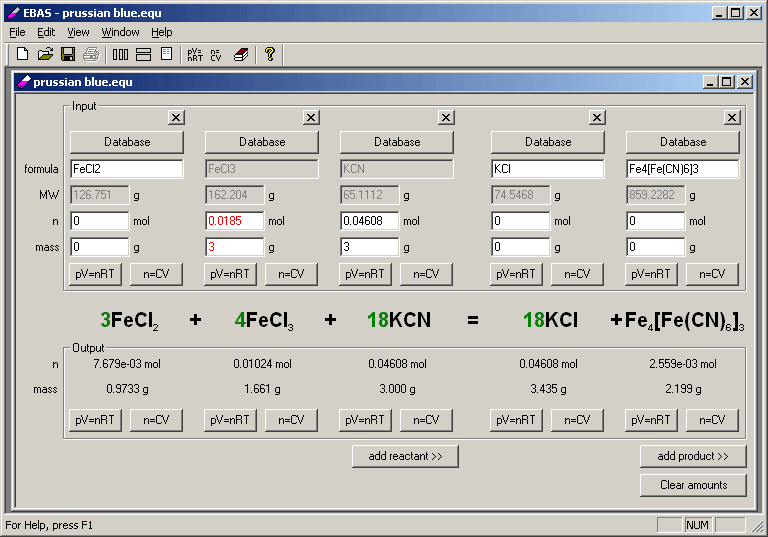
Next, transform the molar equation into a complete ionic equation by dissociating each ionic compound into its respective ions. This balanced chemical equation is the molar equation. 
First, choose any chemical equation that involves ionic compounds and write down its balanced chemical equation. Warning: Lenntech BV cannot be held responsible for errors in the calculation, the program itself or the explanation. The net ionic equation is very easy to find manually. Once you think the equation is balanced, press the Check my answer button. If you have for example Silicates and Fluorides in your water, the judgment on the analysis accuracy is not totally correct. Enter the equation directly into the Balancing Chemical Equations Calculator to balance the given chemical equations. Balance the chemical equations by selecting coefficients from the pull-down menus. Remark: The table above shows only the ions that are usually present in tap water. The chemical equation for a reaction in solution can be written in three ways. The equivalent weight (g/eq or mg/meq) of a compound is defined as: Modified by Joshua Halpern ( Howard University) 12.1: Complete and Net Ionic Equations is shared under a license and was authored, remixed, and/or curated by LibreTexts. The percent difference is calculated with the following formula: Percent difference The principle of electro-neutrality requires that the sum in eq/L or meq/L of the positive ions (cations) must equal the sum of negative ions (anions) in solution, thus: Ions. The calculations are based on the principle of electro-neutrality. This is how the redox equations are balanced. Now, the equation is balanced with 2 Chlorides (Cl) with total charge -2 and 3 Chromiums with total charge +3 on both sides. The acceptance criteria for the percent difference are as given below: This calculator helps you determine the accuracy of your water analysis. To balance the unbalanced chloride molecule charges, we add 2 in front of the chloride on L.H.S. 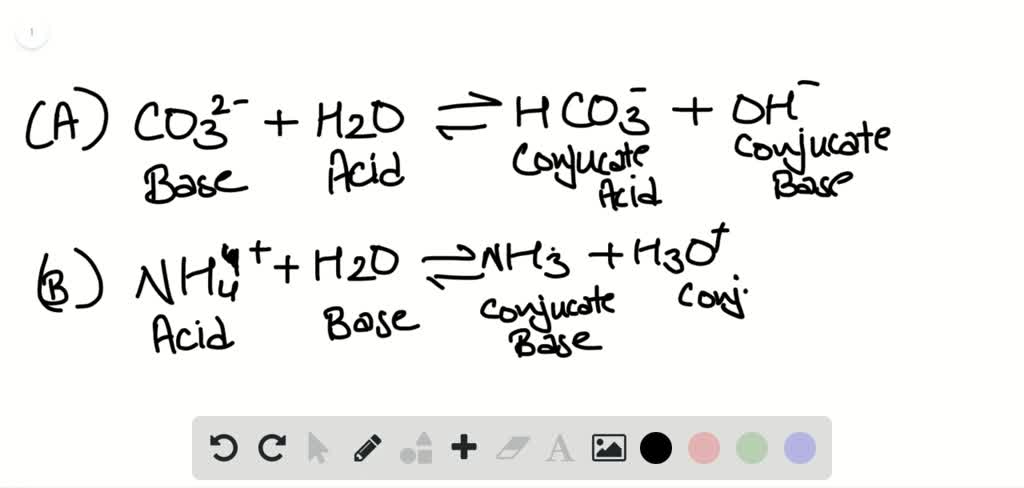
The principle of electro-neutrality requires that the sum in eq/L or meq/L of the positive ions (cations) must equal the sum of negative ions (anions) in solution, thus: The calculations are based on the principle of electro-neutrality. The concentration of acetic acid in the final solution will drop below 0.10 M, but the total of the two species must equal 0.10 M, the initial amount that was put into solution. This calculator helps you determine the accuracy of your water analysis. How to Calculate Faster than a Calculator - Mental Maths 1.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
